 
Recent advances in molecular genetics and imaging technologies have revolutionized diagnosis, counseling for, and treatment of childhood SNHL by precisely defining the etiology and pathology 17, 18. This poses difficulties in the management of auditory neuropathy. Given the heterogeneous etiologies and diverse audiologic features, benefits derived from hearing aids or cochlear implants (CIs) vary significantly among patients 2, 10, 15, 16. Genetic causes of non-syndromic auditory neuropathy include autosomal dominant mutations in DIAPH3 and PCDH9 autosomal recessive mutations in OTOF, PJVK, and GJB2 and mitochondrial mutations in MT-RNR1 9, 11.Īudiologically, auditory neuropathy is characterized by mild to profound SNHL in the presence of otoacoustic emissions (OAEs) and/or cochlear microphonics (CMs), but absent or abnormal auditory brainstem responses (ABRs) and absent acoustic reflexes 2, 9, 10. A number of hereditary neurodegenerative syndromes have been associated with auditory neuropathy, including Charcot-Marie-Tooth disease, Friedreich’s ataxia, Leber’s hereditary optic neuropathy, autosomal dominant optic atrophy, autosomal recessive optic atrophy, Mohr-Tranebjaerg syndrome, Refsum disease, Wolfram syndrome, Pelizaeus-Merzbacher disease, and CAPOS syndrome 9, 11– 14. Genetic factors may contribute to approximately 40% of auditory neuropathy patients in whom clinical manifestations might present as components of specific syndromes or as isolated non-syndromic disorders 9. Auditory neuropathy may also be associated with certain types of syndromic or non-syndromic hereditary hearing impairment 9. Acquired auditory neuropathy include infection during pregnancy, prematurity, kernicterus, and perinatal hypoxia 1, 9. 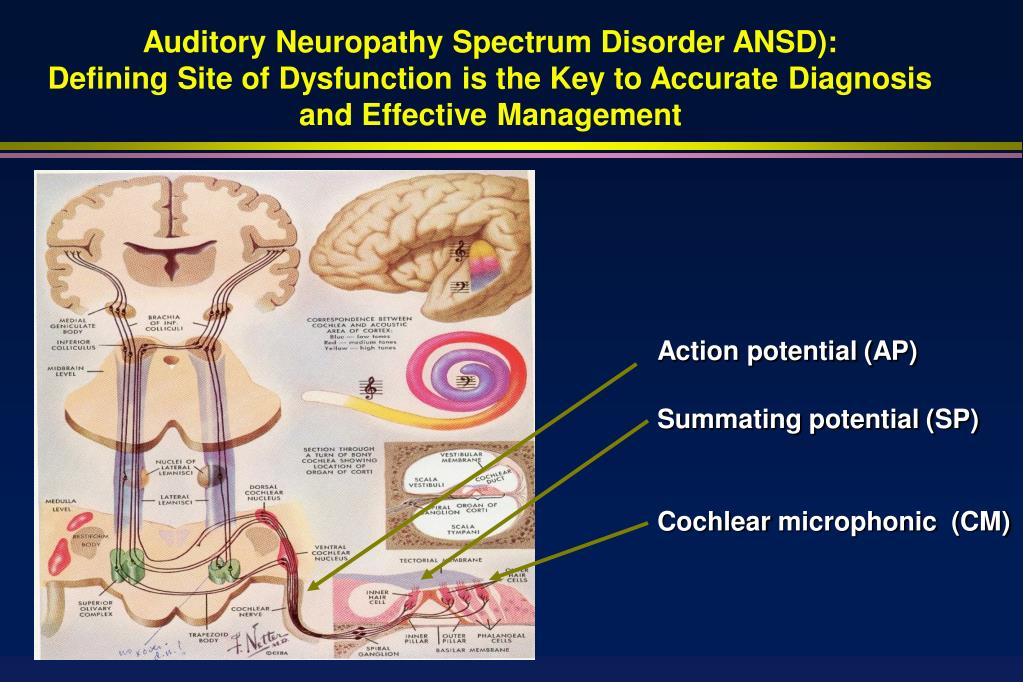
Many acquired and genetic factors can contribute to auditory neuropathy 1, 2, 9, 10. The pathogenesis of auditory neuropathy encompasses a wide range of disease mechanisms, with pathologies localized to multiple sites along the auditory pathway, including inner hair cells 4, synapses 5, spiral ganglion neurons 6, auditory nerve 7, or brainstem auditory nuclei 8. Different etiologies are associated with different audiologic features, and auditory steady-state responses might serve as an objective measure for estimating behavioral thresholds.Īuditory neuropathy is a challenging clinical disorder accounting for ~10% of cases of sensorineural hearing loss (SNHL) in children 1– 3. In conclusion, comprehensive assessments can provide etiological clues in ~75% of the children with auditory neuropathy. In patients with different etiologies or pathological sites, moderate to strong correlations (Pearson’s r = 0.51–0.83) were observed between behavioral thresholds and auditory steady-state response thresholds. Patients with acquired auditory neuropathy presented hearing loss earlier (odds ratio, 10.2 95% confidence interval, 2.2–47.4), whereas patients with genetic auditory neuropathy had higher presence rate of distortion product otoacoustic emissions (odds ratio, 10.7 95% confidence interval, 1.3–85.4). The most common causes of acquired and genetic auditory neuropathy were prematurity and OTOF mutations, respectively. Etiologically, 48 (47.5%), 16 (15.8%), 11 (10.9%), and 26 (25.7%) children were categorized as having acquired, genetic, cochlear nerve deficiency-related, and indefinite auditory neuropathy, respectively. 
This study used an integrative patient-history, audiologic, genetic, and imaging-based approach to investigate the etiologies and audiologic features of 101 children with auditory neuropathy. Due to diverse etiologies and clinical features, the management is often challenging. Auditory neuropathy is an important entity in childhood sensorineural hearing loss. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
